Determinación experimental de las propiedades estructurales y mecánicas en la mitad de la diáfisis del hueso fémur de canino sometido a ensayo de flexión
Resumen
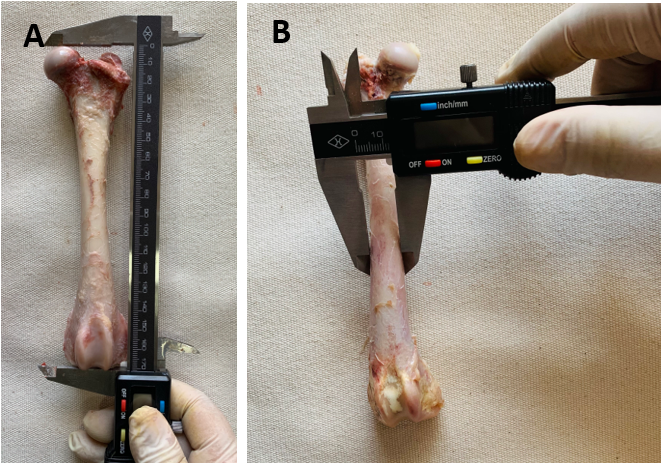
Las características estructurales de los huesos son influenciadas por la especie, edad y sexo del animal. La determinación experimental de propiedades mecánicas del tejido óseo en caninos es fundamental para la ortopedia y la traumatología. Este trabajo pretende determinar las propiedades mecánicas y estructurales del tejido óseo cortical en huesos fémur de perro, aportando conocimientos integrales y brindando información con finalidad clínico-quirúrgica para la práctica de la Medicina Veterinaria. Se procedió a evaluar la resistencia del tercio medio de la diáfisis de los huesos fémures mediante ensayo de flexión en tres puntos, la cual se relacionó con las variables morfológicas óseas. Además, se realizó la comparación entre la resistencia del tercio medio de la diáfisis del fémur de perros de diferente talla, edad y sexo. Los valores promedio obtenidos fueron: fuerza máxima 0,96 kN ± 0,12, deformación 5,95 mm ±1,10. Los huesos fémur de animales de talla grande presentaron una resistencia significativamente mayor ante la flexión que los de talla chica (p=0,0124). Los fémures de perros machos presentaron una mayor resistencia que aquellos provenientes de hembras (p=0,017). Al aumentar la edad del animal el hueso tuvo una menor deformación ante la flexión en tres puntos (p= 0,029). Se concluye que un alto porcentaje (76 %) de la variabilidad de la fuerza máxima, soportada por el hueso antes de la fractura, puede explicarse por los valores de área cortical.
Descargas
Citas
Beale B. (2004). Techniques for the Management of Long Bone Fractures Orthopedic clinical techniques femur fracture repair. Clinical Techniques in Small Animal Practice. 19 (3): 134-150.
Calvo Soler P. (2017). Vínculos entre el ser humano y los animales: aspectos psicológicos y psicopatológicos. Tesis Doctoral. Universidad Autónoma de Barcelona.
Coll Cárdenas F, Olivera D. (2018). Biofísica para estudiantes de Ciencias Veterinarias. Editorial Universidad Nacional de La Plata. 1° Edición. Capítulo 6. 109.
Di Rienzo JA, Casanoves F, Balzarini M.G, González L, Tablada M. (2018). InfoStat versión 2018.Grupo InfoStat, Facultad de Ciencias Agrarias, Universidad Nacional de Córdoba, Argentina.
Fioretti C, Natali J, Galán A, Rivera MC, Moine R, Varela P, Varela M, Bonino F, Quinteros R. (2011). Características Mecánicas Dinámicas del Fémur Aislado de Perro, Sometido Prueba de Impacto. International Journal of Morphology Chile. 29: 716-722.
Fioretti C, Galán A, Moine R, Varela M, Varela P, Mouguelar H, Gigena S, Bonino F, Quinteros R, Natali J. (2013). Características Mecánicas Dinámicas de la Tibia Aislada de Perro Sometida a Prueba de Impacto. International Journal of Morphology. Chile. ISSN 0717 – 9502. 31 (2): 562-569.
Fioretti RC, Moine R, Varela M, Quinteros R, Varela P, Galán AM, Gigena S, Mouguelar H, Gonzalez Sanchez S, Natal N. (2018). Densidad mineral ósea y resistencia ante la prueba de compresión en la mitad de la diáfisis del hueso fémur de perro. Ab Intus. ISSN 1234-5678. Volumen 1 (1): 43-52.
Fioretti RC, Moine R, Varela P, Gigena S, Salvi M, Audap Soubie R, de Prada R, Giorgetti L, Varela M, Gonzalez Sanchez S, Cancino M, Nozzi M, Comeglio G, Boatti J. (2022). Emisión acústica y prueba de flexión en tres puntos en la diáfisis de la falange proximal del dedo de la mano de equinos mestizos criollos. Ab Intus. ISSN 2618-2734. 10 (5): 25-32.
Koscinczuk P. (2017). Domesticación, bienestar y relación entre el perro y los seres humanos. Revista veterinaria 28: 1, 78-87.
León M, Ramírez EI, Ruiz O, Jacobo VH. (2016). Análisis de la rigidez de un fémur a partir del modelado de un ensayo de flexión en cuatro puntos. Memorias del XXII Congreso de la SOMIM y XIV Congreso Iberomat CD. Mérida, Yucatán. México.
Moine R, Rivera C, Vivas A, Ferraris, G, Galán A, Natali J. (2001). Morfometría y determinación de calcio y fósforo en la parte media de la diáfisis del metacarpiano III en yeguas mestiza con criollo. Archivos de Medicina Veterinaria. Chile. ISSN 0301 – 732X. XXXIII, N° 1: 63 - 68.
Moine R, Galán M, Vivas A, Fioretti C, Varela M, Bonino F, Quinteros R, Natali J. (2015). Propiedades Morfológicas en la Parte Media de la Diáfisis del Hueso Metacarpiano III de Equino Mestizo Criollo. International Journal of Morphology. Chile. ISSN 0717 – 9502. 33 (3): 955-961.
Moine R, Fioretti R, Galan A, Gigena S, Salvi M, Audap Soubie R, Varela P, Varela M, Gonzalez Sanchez S, Natali J. (2020). Propiedades estructurales y resistencia a la flexión en tres puntos en la parte media de la diáfisis de la falange proximal de la mano del caballo. Ab Intus. 56-62. ISSN 2618-2734. 6 (3): 47-56.
Natali J, Wheeler JT, Kohl R, Varela P. (2008). Comparación de las Características Mecánicas Estáticas del Fémur Aislado de Perro, con y sin la Colocación de una Placa de Ortopedia Fabricada en polipropileno. International Journal of Morphology. Chile. ISSN 0717 – 9502. 26(4): 791-797.
Natali J, Fioretti R, Moine R, Gigena M, Mouguelar H, Varela M, Gonzalez Sanchez S, Quinteros R, Galán A. (2019). Morfología y comportamiento biomecánico de la falange proximal de la mano del caballo mestizo criollo. Ab Intus. ISSN 2618-2734. 3 (2): 56-62.
Ocampos M, Penagos E, Ramírez N. (2011). Tratamiento de una fractura distal del Fémur de un canino joven. Ciencias Agropecuarias. 3 (2):40-43.
Orozco Santos P, Ruiz GL, Rivera Montoya GF, Jaramillo Rivas J. (2017). Valores de densidad Mineral Ósea (DMO) y Densidad Mineral Volumétrica (DMOV) a Nivel de la Columna Lumbar, en adolescentes y adultos jóvenes. Revista de Investigaciones ALETHEIA Número 7 Corporación Universitaria Empresarial Alexander von Humboldt. 143 – 155.
Pellegrino F y Bruzzo Lafratto J. (2018). Biofísica del aparato locomotor animal Capítulo 6.113-123. https://ri.conicet.gov.ar/bitstream/handle/11336/113465/CONICET_Digital.
Varela FM, Fioretti RC, Natali J, Moine RM, Quinteros R, Galán AM. (2019). Morfometría y resistencia de los huesos del brazo y antebrazo del perro. https://www.hum.unrc.edu.ar/wp-content/uploads/2019/10/2-biolog1.pdf. Edición especial. Revista Científica Facultad de Agronomía y Veterinaria, Universidad Nacional de Río Cuarto. Ab Intus. ISSN 2618-2734. 2, (4): 122.
Varela M, Moine R, Fioretti RC, Varela P, de Prada R, Giorgetti L, Bellingeri R, Barbero. (2021). Análisis mecánico y morfométrico del hueso carpo III del caballo mestizo Criollo Argentino. Revista de Medicina Veterinaria (En línea), ISSN 1852-771X. 102 (2): 36-42.
Wheeler JT, Adagio L, D Amico G, Hierro J, Hagge M, Lattanzi D, Schieda F, Sanfilippo S. (2002). Fracturas de huesos largos en caninos inmaduros. Ciencia Veterinaria. Facultad de Ciencias Veterinarias Universidad Nacional de La Pampa. Página 57-67.
Publicado
Cómo citar
Número
Sección
Licencia
Derechos de autor 2023 Ab Intus

Esta obra está bajo una licencia internacional Creative Commons Atribución-NoComercial 4.0.

















